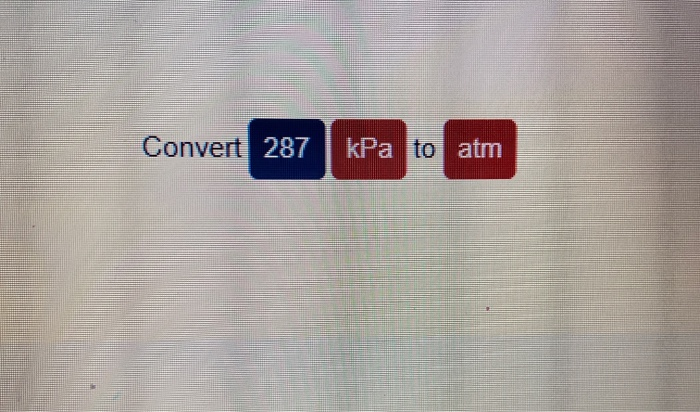
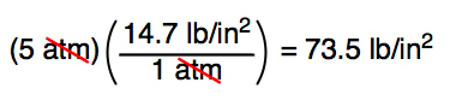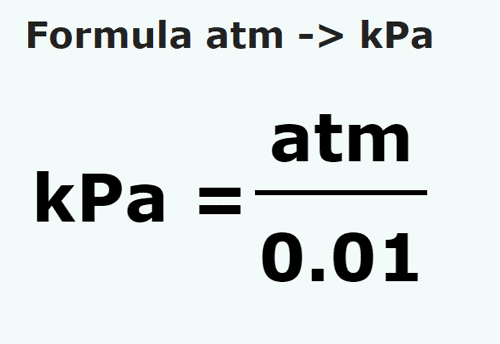✓ Solved: Gas pressures can be expressed in units of mm Hg, atm, torr, and kPa. Convert these pressure...
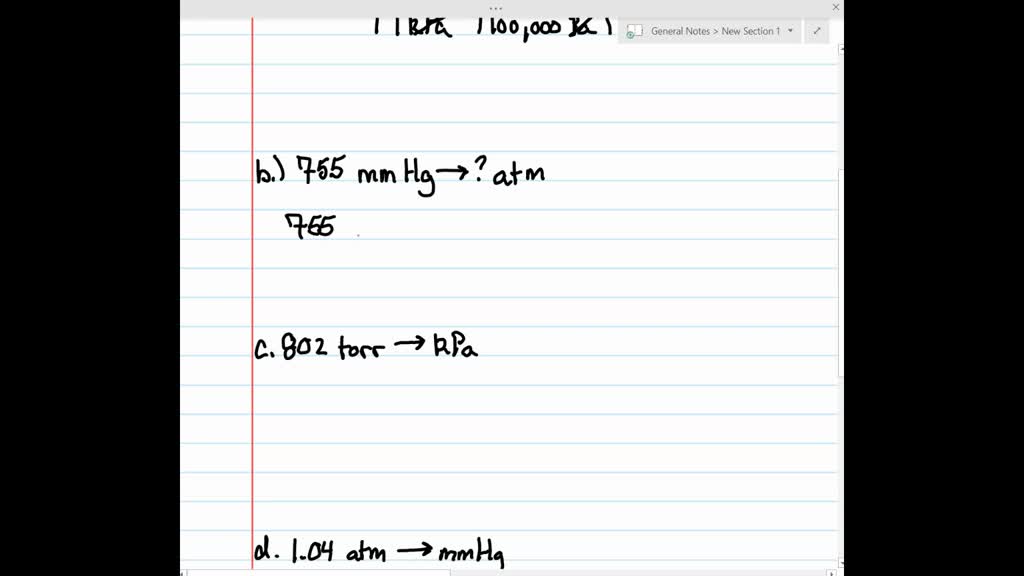
SOLVED:Make the indicated pressure conversions. a. 45.2 kPa to atmospheres b. 755 mm Hg to atmospheres c. 802 torr to kilopascals d. 1.04 atm to millimeters of mercury
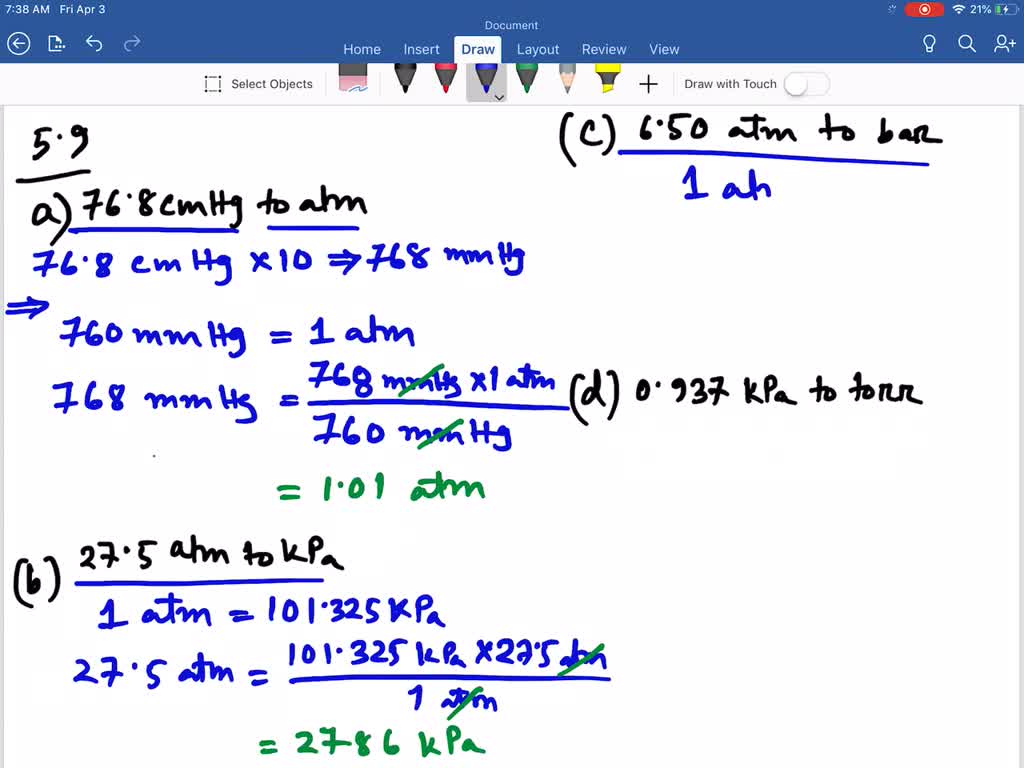
SOLVED:Convert the following: \begin{array}{ll}{\text { (a) } 76.8 \mathrm{cmHg} \text { to atm }} & {\text { (b) } 27.5 \text { atm to kPa }} \\ {\text { (c) } 6.50 \